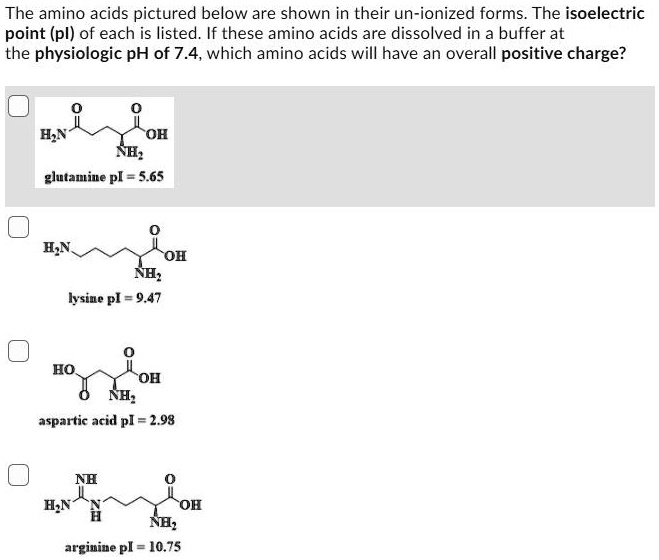
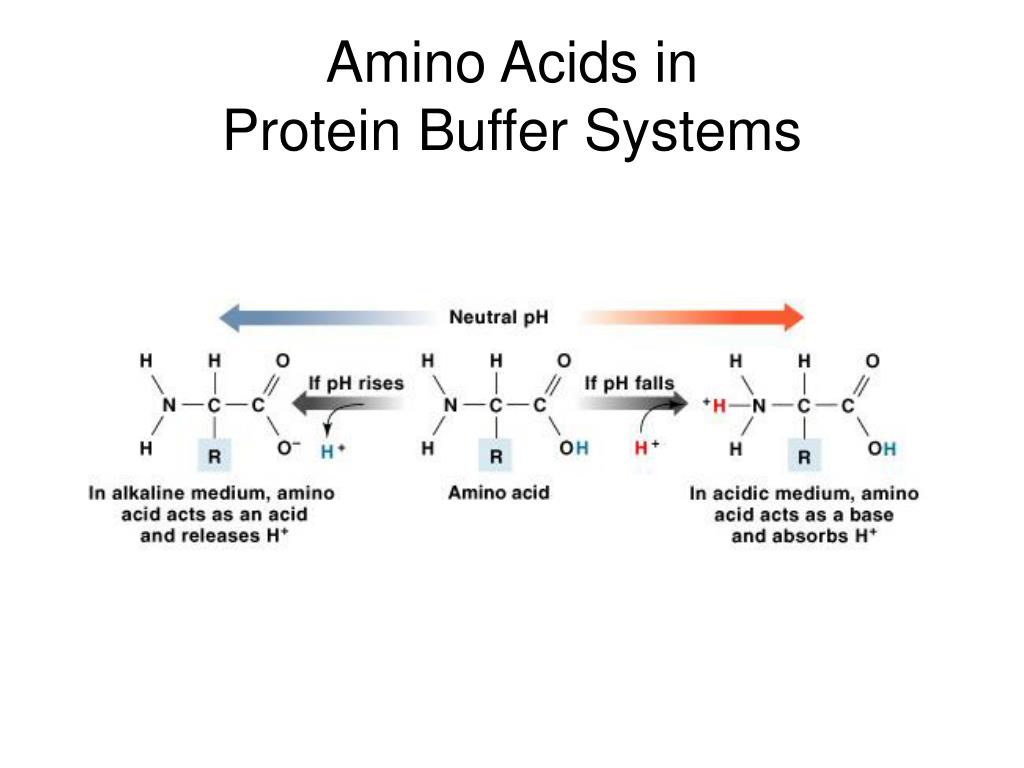
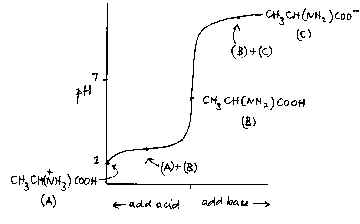
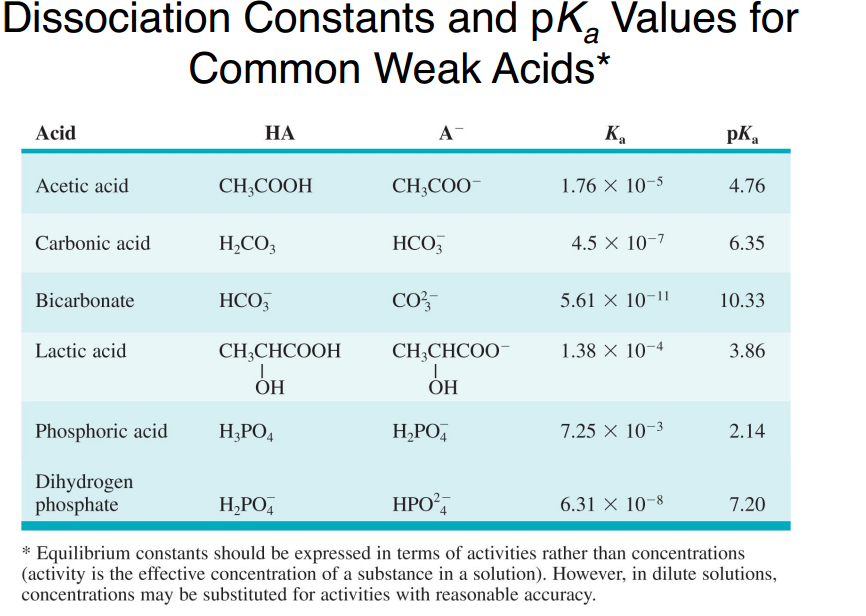
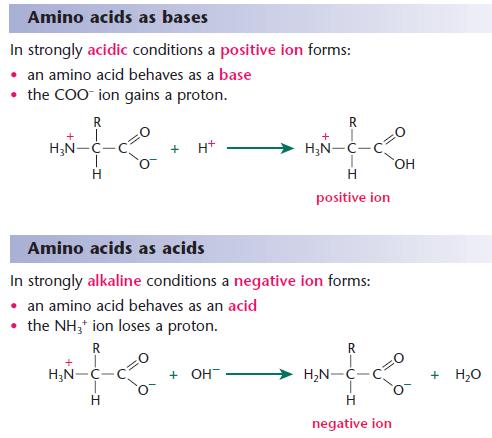
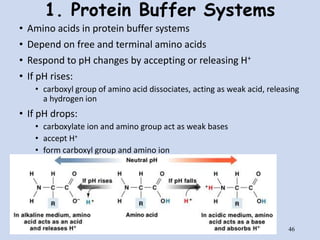
![Buffer for High-Speed Amino Acid Analyzer PH-SET・028-19511[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation Buffer for High-Speed Amino Acid Analyzer PH-SET・028-19511[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/sc/05/028-19511.png)
Buffer for High-Speed Amino Acid Analyzer PH-SET・028-19511[Detail Information] | [Analytical Chemistry]|Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation

Comparison of three buffer solutions for amino acid derivatization and following analysis by liquid chromatography electrospray mass spectrometry. | Semantic Scholar

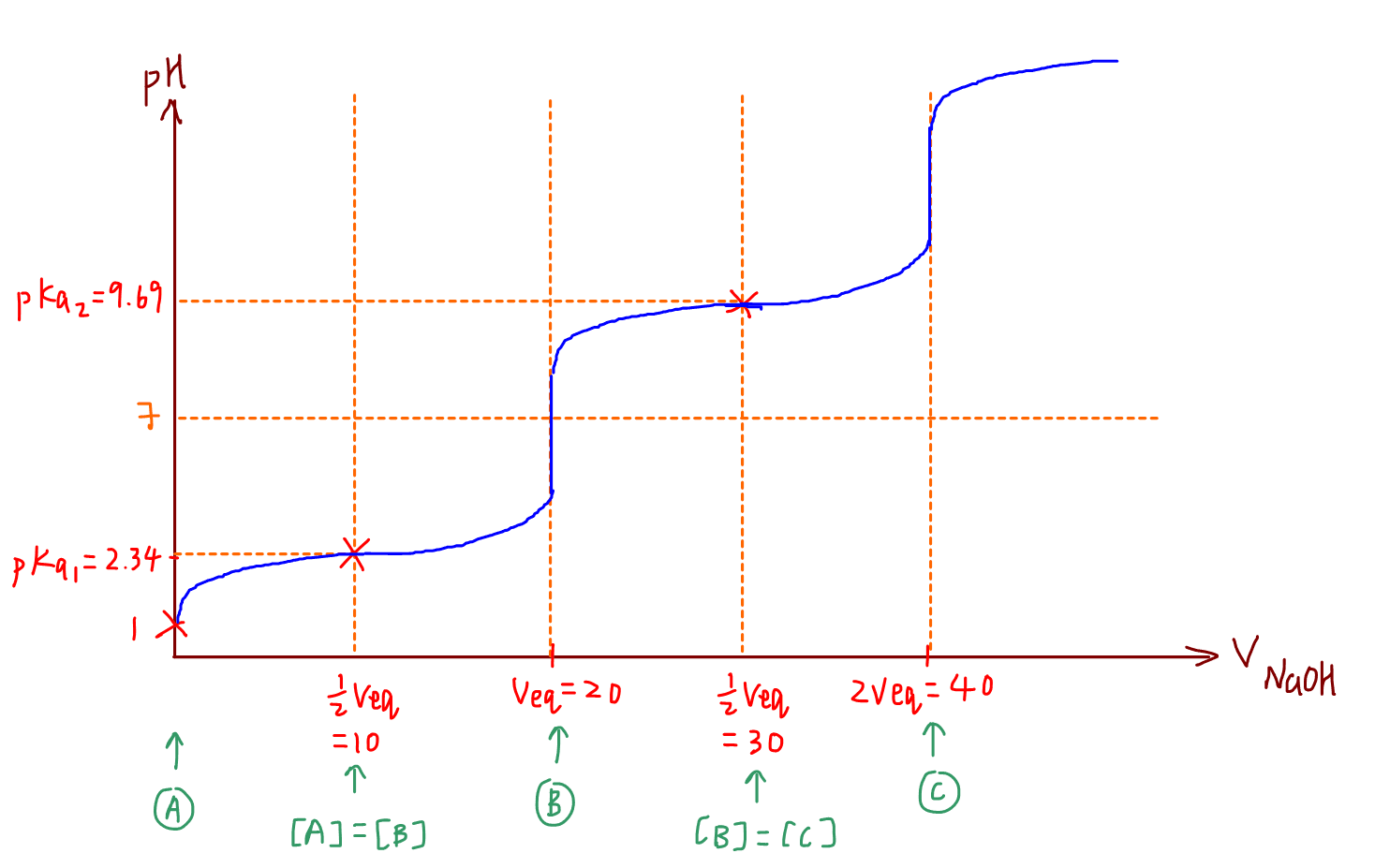
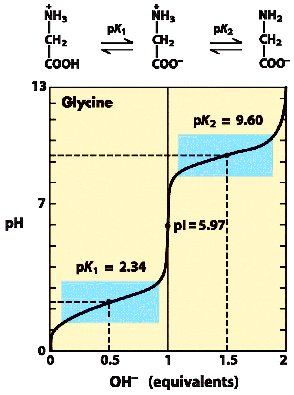
A buffer using the amino acid glycine is prepared by mixing 80 ml of 1.0M glycine, pH 0.5 (i.e. fully protonated), 55 ml of 2M NaOH and 365 ml of water. Calculate

Understanding the Stabilizing Effect of Histidine on mAb Aggregation: A Molecular Dynamics Study | Molecular Pharmaceutics