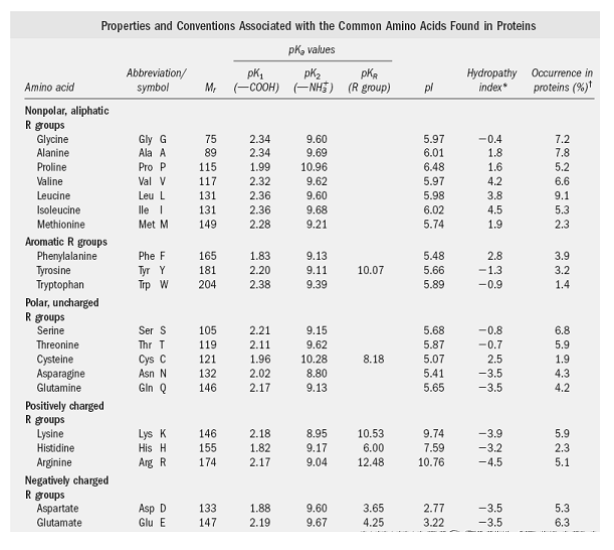
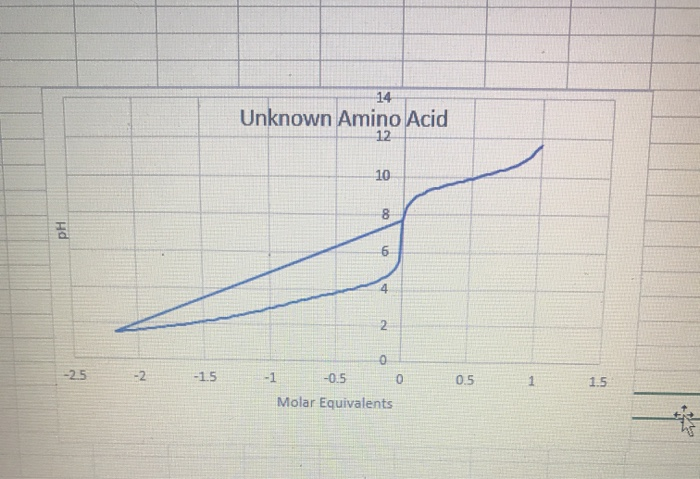
SOLVED: What is the pI of the polypeptide DRAW? The pKa values for the free amino acids are pK1, which refers to the pKa of the COOH group, pK2, which refers to
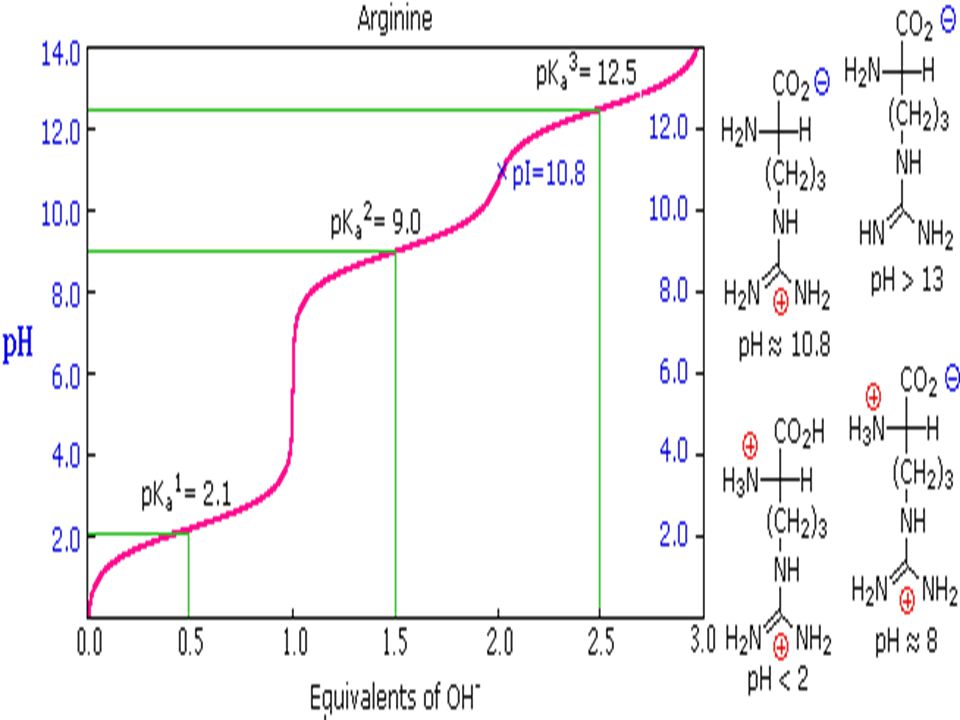
The pK1, pK2, and pKR for the amino acid arginine are 1.8, 9.0, and 12.5 respectively. At pH 7.0 arginin would be charged predominantly as what? - Quora
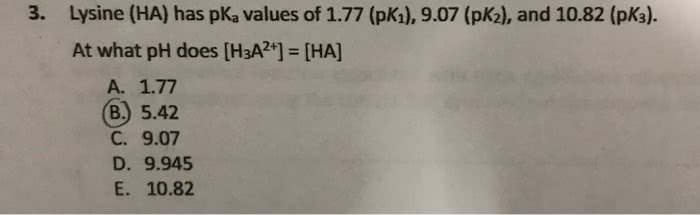
OneClass: 3. Lysine (HA) has pKa values of 1.77 (pKı), 9.07 (pK2), and 10.82 (pK3). At what pH does ...
SOLVED: The pK1, pK2, and pKR of the amino acid lysine are 2.2, 9.1, and 10.5, respectively. The pK1, pK2, and pKR of the amino acid arginine are 1.8, 9.0, and 12.5,

The amino acid methionine has pKa1 = 2.2 and pKa2 = 9.1. If this amino acid is represented by H2L+, what is the major species at pH 6? | Homework.Study.com

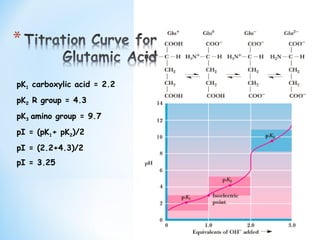
The pKa1 and pKa2 of an amino acid are 2.3 and 9.7 respectively. The isoelectric point of the amino acid is:
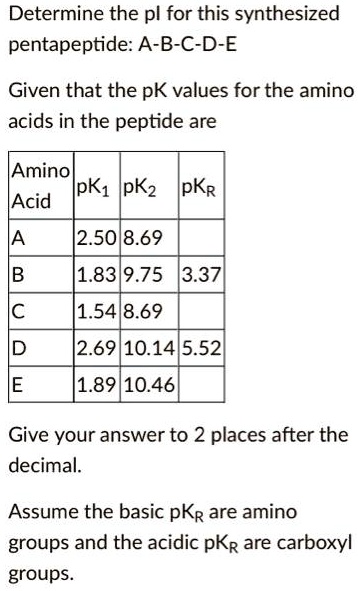
SOLVED: Determine the pI for this synthesized pentapeptide: A-B-C-D-E Given that the pK values for the amino acids in the peptide are: Amino acid pK1 pK2 pK3 Acidic pKR Basic pKR A
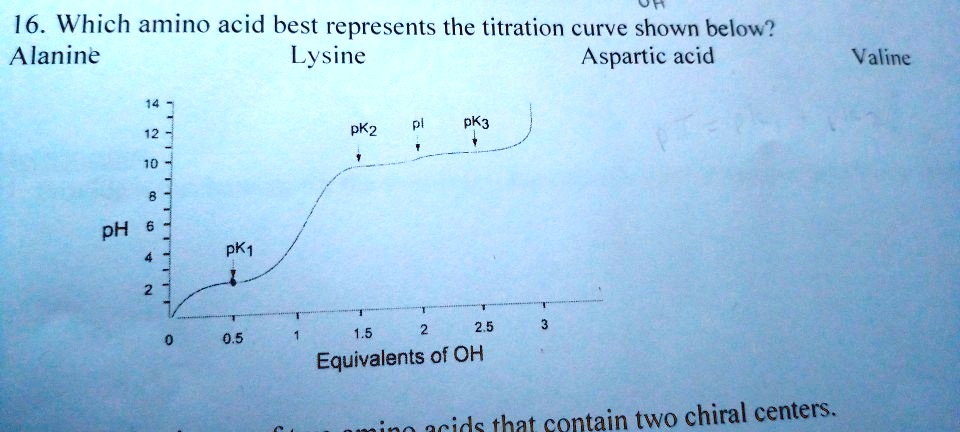
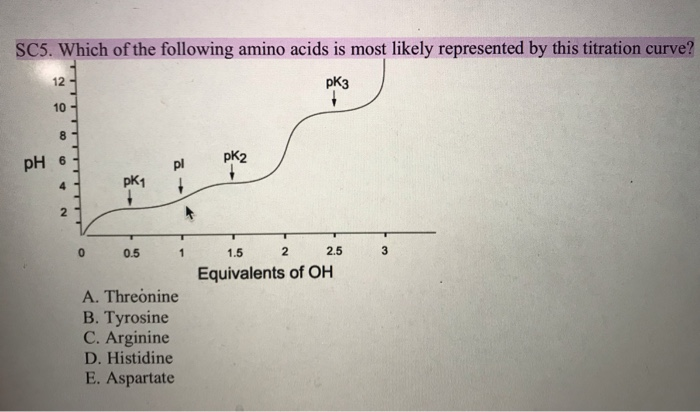
SOLVED: Ot 16. Which amino acid best represents the titration curve shown below? Alanine Lysine Aspartic acid Valine Exd Zxd Hd PK1 1.5 2,5 Equivalents of OH idc that contain two chiral centers.