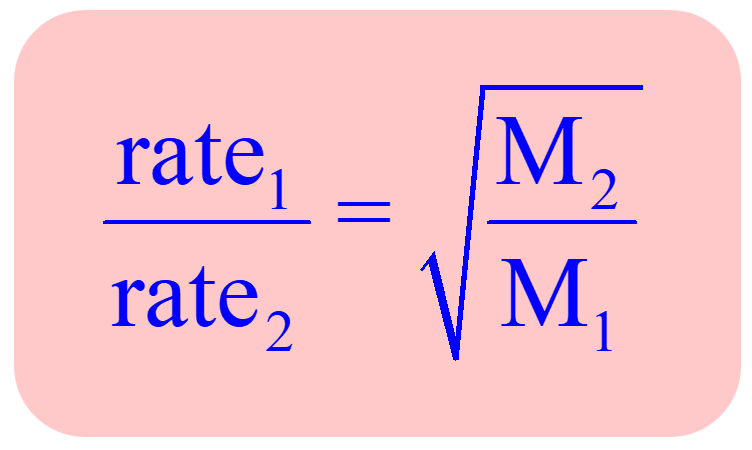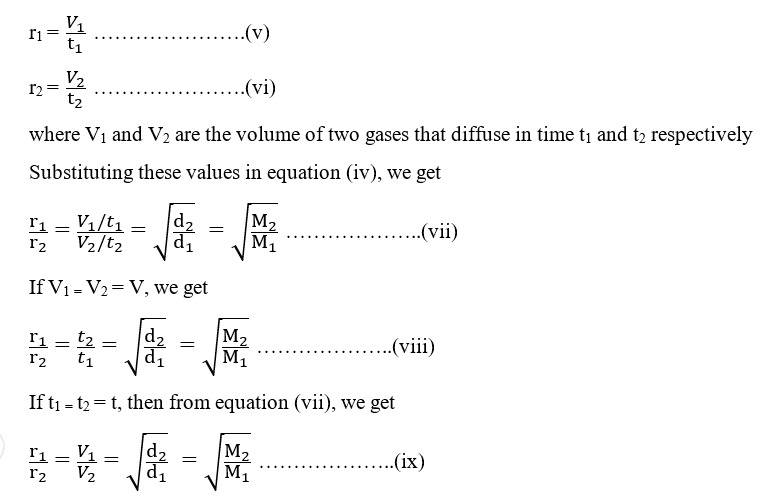Can the rate of effusion or diffusion be negative, in accordance with Graham's law? If so, how? - Quora
Rate of diffusion of a saturated hydrocarbon is about 1/6 th of that of hydrogen under similar conditions of temperature and pressure. What is the molecular formula of that hydrocarbon?

Graham's Law of Diffusion Graham's Law KE = ½mv 2 Speed of diffusion/ effusion –Kinetic energy is determined by the temperature of the gas. –At the same. - ppt download