What does it mean when orbitals are called sharp (s orbital); principle (p orbital); diffuse (d orbital) and fundamental (f orbital)? - Quora

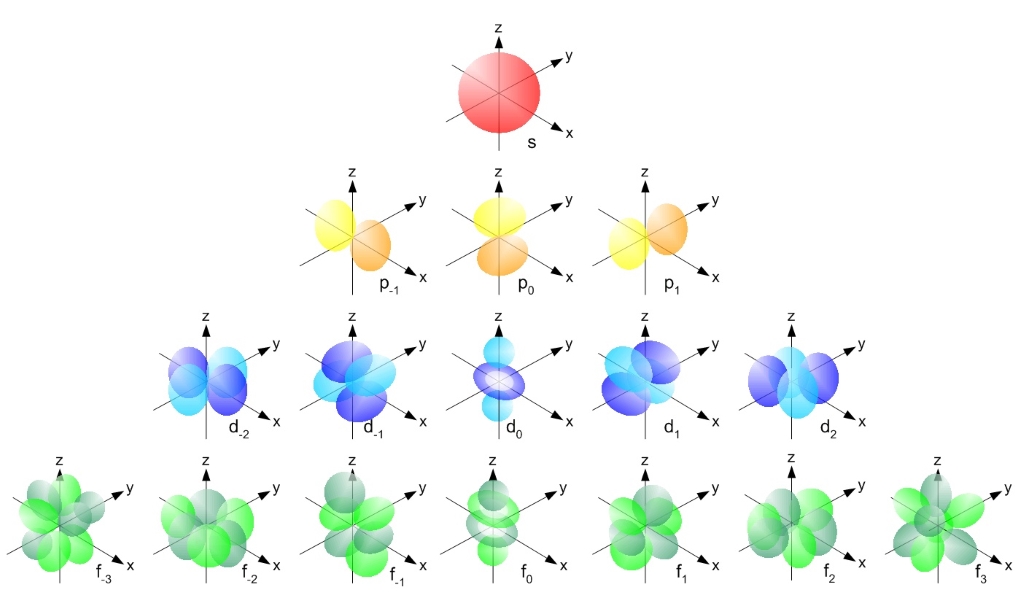
s,p,d,f Orbitals - Chemistry | Socratic | Chemistry, Physics and mathematics, Electron configuration
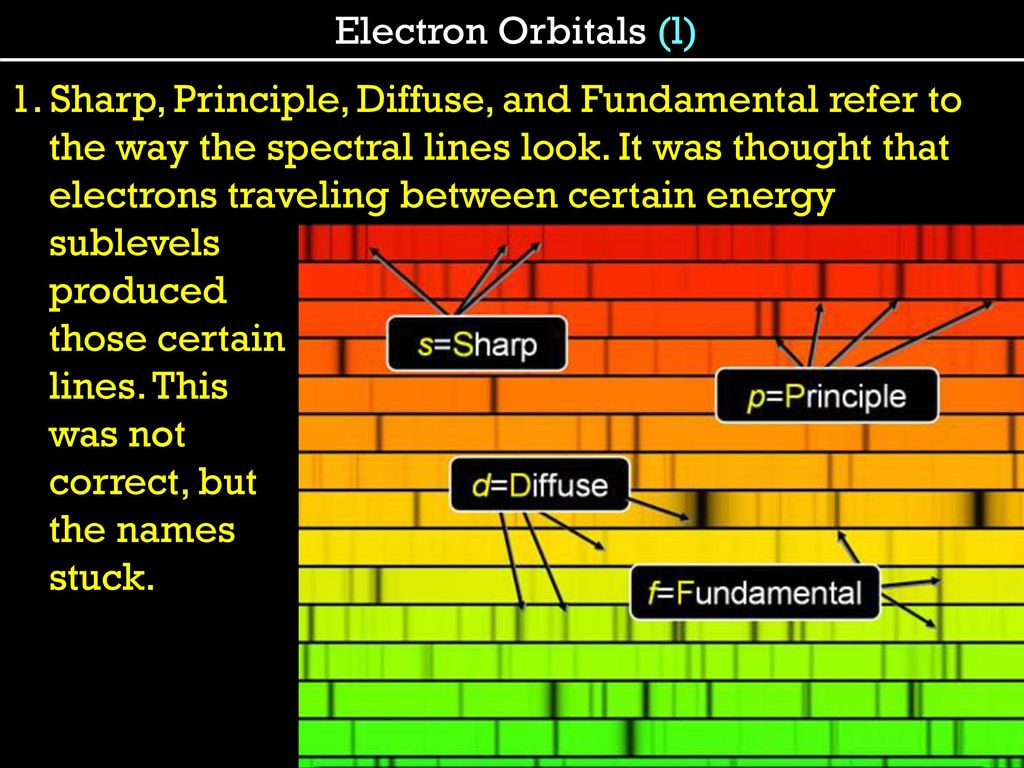
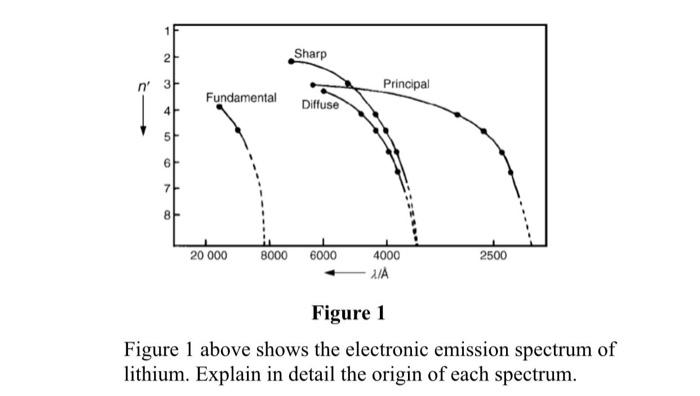
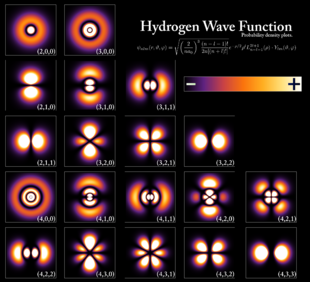
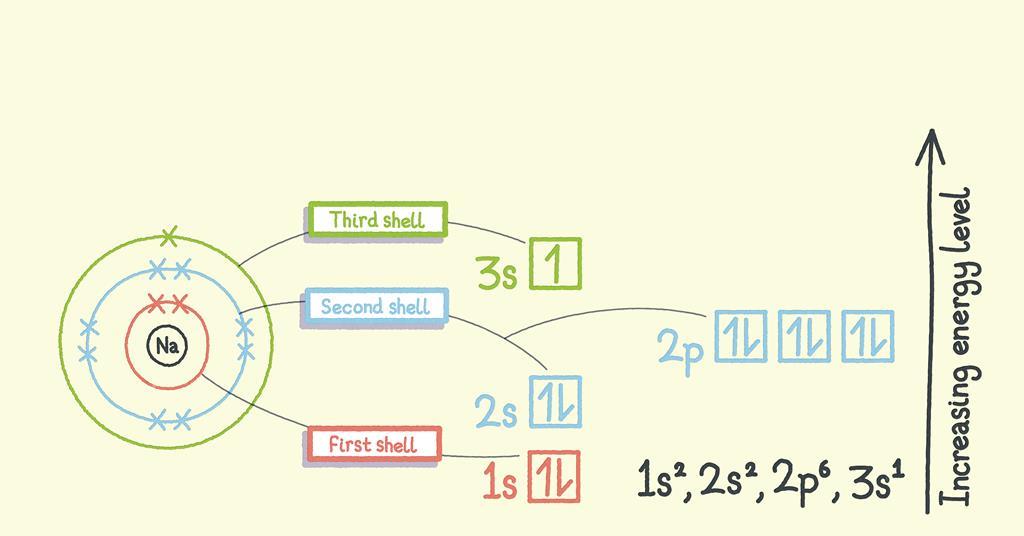
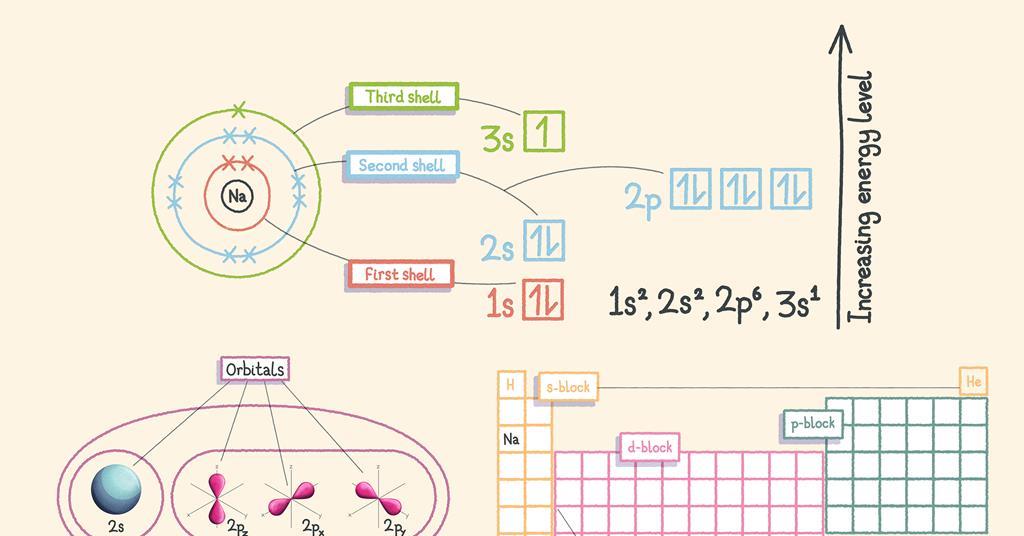
The subsidiary/azimuthal quantum number is s, p, d, f, g, h and so on. where the s, p, d, and f stand for sharp, principal, diffuse and fundamental, respectively. What is the